A groundbreaking study reveals a critical process in the aging brain that may explain why the risk of neurodegenerative diseases like Alzheimer’s increases with age: the slowdown of protein recycling. This isn’t just about general wear and tear; it’s about how brain cells handle their waste, and when that system breaks down, the consequences can be severe.
The Breakdown of Cellular Cleanup
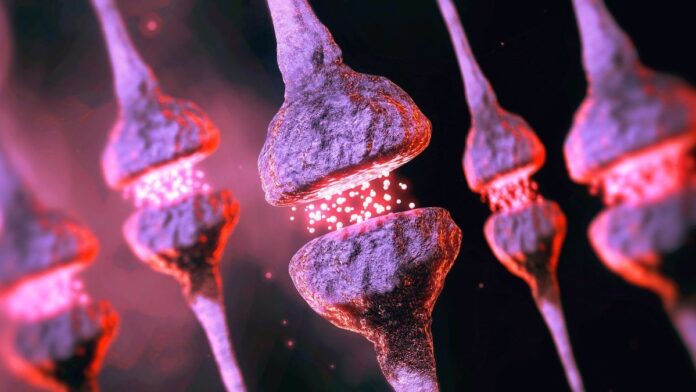
As we age, neurons become less efficient at breaking down and recycling old proteins. This leads to a buildup of clumpy, misfolded proteins – particularly at synapses, the vital connections between brain cells. Instead of being cleared away, these proteins are offloaded onto other cells, overwhelming the brain’s natural cleanup crew, microglia.
This is more than just inefficiency; it’s a fundamental shift in how the brain manages its biological waste. The brain has over a quadrillion synapses, each maintained by synaptic proteins, and their failure to recycle efficiently creates a toxic buildup over time. The study, conducted on mice, showed that aged brains took twice as long to recycle neuronal proteins, allowing these clumps to accumulate and disrupt cell function.
Microglia: Overwhelmed Guardians
Microglia are the brain’s immune cells, responsible for clearing debris and pruning damaged synapses. When overloaded with protein clumps, they become overwhelmed, potentially becoming dysfunctional themselves. This creates a vicious cycle: the cleanup crew breaks down, further accelerating the buildup of waste and damaging healthy brain connections.
This is critical because synaptic loss is directly linked to cognitive decline and neurodegenerative diseases. While many factors contribute to these conditions, this study highlights a core mechanism: the inability of neurons to efficiently process and eliminate cellular waste.
New Avenues for Research
Researchers tagged and tracked thousands of proteins in mice of different ages, using a novel biological tag that revealed how long proteins lasted before being broken down. The results showed that the decline in protein recycling emerged after middle age, suggesting a specific window of vulnerability.
The study also identified proteins previously unknown to be linked to neurodegeneration, opening new avenues for biomarker discovery and therapeutic targets. The ability to monitor protein degradation in living cells is a significant technical advancement, allowing for more precise study of brain health during aging.
“If we can leverage our system to study neuron-derived proteins in the blood during aging and disease, we could potentially identify new biomarkers of brain health,” explains Ian Guldner, lead researcher at Stanford University.
The Bigger Picture
This research doesn’t offer a cure for Alzheimer’s, but it provides a crucial piece of the puzzle. Understanding how the brain’s ‘garbage disposal’ system fails with age could lead to interventions that slow down protein buildup, protect microglia, and preserve cognitive function. The findings underscore the importance of cellular maintenance in long-term brain health and highlight the need for further research into age-related neurodegeneration.